RD-E: 4300 Perfect Gas Modeling with Polynomial EOS
Polynomial EOS is used to model perfect gas. Pressure or energy can be absolute values or relative. Material LAW6 (/MAT/HYDRO) is used to build material cards for each of these cases.
Figure 1.
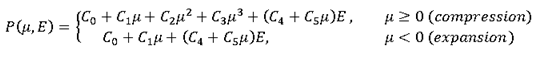
| Case | Mathematical Model | Pressure | Energy |
|---|---|---|---|
| 1 | absolute | absolute | |
| 2 | relative | absolute | |
| 3 | relative | relative | |
| 4 | absolute | relative |
A simple test of compression/expansion is made to compare these formulation outputs with theoretical results.
Options and Keywords Used
- Perfect gas
- Polynomial EOS (/EOS/POLYNOMIAL)
- Absolute / Relative formulations
- Pressure shift
- Hydrodynamic fluid material (/MAT/LAW6 (HYDRO or HYD_VISC))
- Imposed displacement (/IMPDISP)
Nodes on each of the faces are moved with imposed displacement
- Boundary conditions (/ALE/BCS)
Boundary nodes are defined as Lagrangian
Element pressure, density and internal energy density are saved in the Time History file.
Input Files
- Model 1
- <install_directory>/hwsolvers/demos/radioss/example/43_perfect_gas_polynomial_eos/01-Pabsolute_Eabsolute/*
- Model 2
- <install_directory>/hwsolvers/demos/radioss/example/43_perfect_gas_polynomial_eos/02-Prelative_Eabsolute/*
- Model 3
- <install_directory>/hwsolvers/demos/radioss/example/43_perfect_gas_polynomial_eos/03-Prelative_Erelative/*
- Model 4
- <install_directory>/hwsolvers/demos/radioss/example/43_perfect_gas_polynomial_eos/04-Pabsolute_Erelative/*
Model Description
Figure 2.
- 1e5 Pa
- 1000 m3
- 1.204
- 0
The fluid will be assumed to be a perfect gas. Volume is changed in the three directions to consider a pure compression followed by an expansion of matter (Figure 3).
This test will be modeled with a single ALE element (8 node brick) and polynomial EOS.
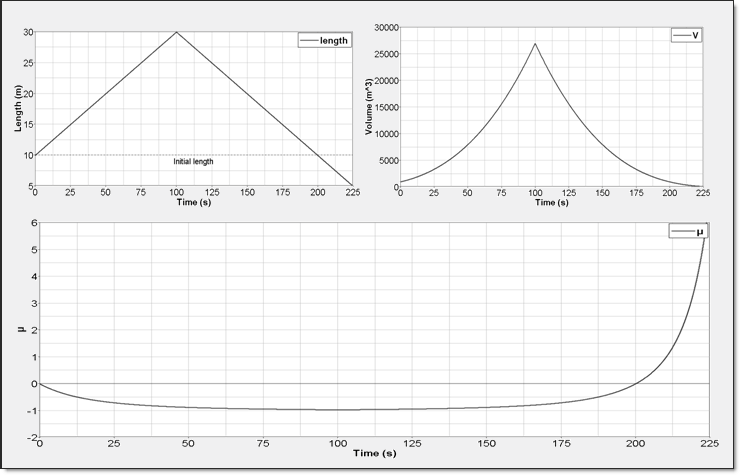
Figure 3. Elementary Volume Change. Length is Modified with /IMPDISP Card; its Influences on and are Plotted
Polynomial EOS
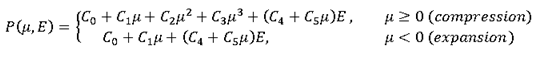
- Hydrodynamic pressure
- General case corresponds to Mie-Guneisen EOS (refer to Appendix B: Basic Relations of Elasticity of the Theory Manual)
- Incompressible gas
- Linear elastic material
- Perfect gas
This example is focused only on Perfect Gas modeling.
Model Method
A single ALE brick element is used. Material is confined inside the element by defining brick nodes as Lagrangian. For each face, displacement is imposed on the four nodes along the normal.
| (1) | (2) | (3) | (4) | (5) | (6) | (7) | (8) | (9) | (10) |
|---|---|---|---|---|---|---|---|---|---|
| /MAT/LAW6/mat_ID/unit_ID or /MAT/HYDRO/mat_ID/unit_ID | |||||||||
| mat_title | |||||||||
| C0 | C1 | C2 | C3 | ||||||
| Pmin | Psh | ||||||||
| C4 | C5 | E0 | |||||||
Pressure Shift
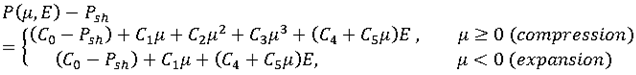
Radioss Engine shifts C0 flag and computed pressure with an offset of -Psh.
Minimum Pressure
The theoretical value is (absolute pressure) with a default value of -1030, to accept a negative value in relative pressure formulation.
This flag has to be manually offset with -Psh.
Results
Theoretical Results
- PressurePerfect gas pressure is given by:
(9) Then,(10) Radioss assumes the hypothesis of an isentropic process to compute the change in internal energy:(11) This theory gives the following differential equation:(12) This has the form and the general solution is:(13) Pressure is also polytropic:(14) (15) Here, is the material constant (ratio of heat capacity). For diatomic gas =1.4. Air is made mainly of diatomic gas, so set gamma to 1.4 for air.
- Internal Energy
- Sound SpeedPerfect gas sound speed is:
(17) Equation 15 gives its expression in term of volume:(18)
| Pressure (Pa) | Internal Energy Density (J) | Sound Speed (m/s) | |||
|---|---|---|---|---|---|
| PREF(V) | PREF() | eREF(V) | eREF() | cREF(V) | cREF() |
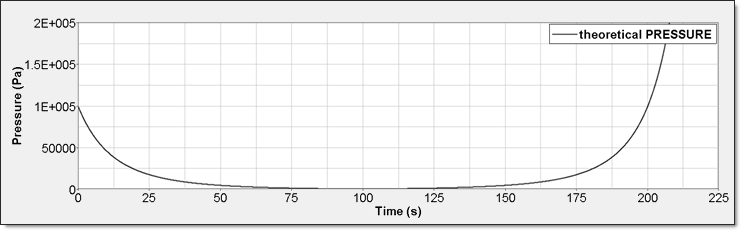
Figure 4. Perfect Gas Pressure
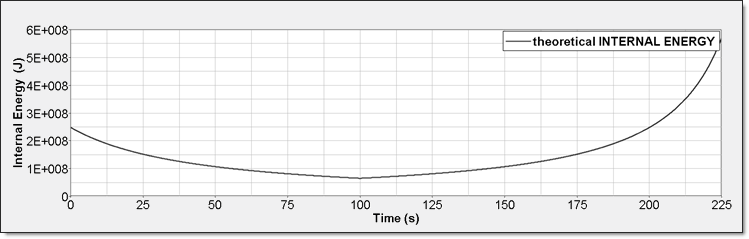
Figure 5. Perfect Gas Internal Energy
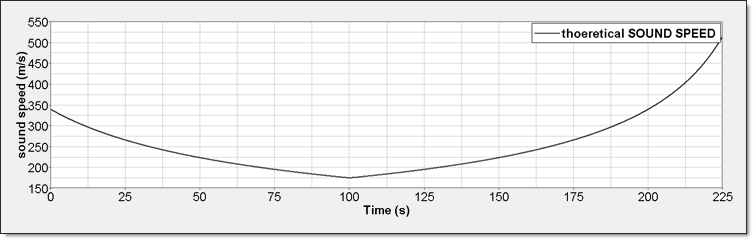
Figure 6. Perfect Gas Sound Speed
Material Control Cards
- Case 1: Both Pressure and Energy are absolute values:
- Case 2: Pressure is relative and Energy is absolute:
- Case 3: Both Pressure and Energy are relative:
- Case 4: Pressure is absolute and Energy is relative:
Sound Speed and Time Step
| Case | C0 | C1 | C4 | C5 | c2 | Comparison with Theoretical Value |
|---|---|---|---|---|---|---|
| 1 | 0 | 0 | c = cREF | |||
| 2 | 0 | 0 | c = cREF | |||
| 3 | c = cREF | |||||
| 4 | c = cREF |
For each of the four formulations, the computed sound speed by Radioss is the same as the theoretical one. Time step and cycle number are also not affected.
Case 1: Both Pressure and Energy are Absolute Values
- Equation of StateEquation of state can be written:
(34) with(35) Expanding this expression and identifying the polynomial coefficients leads to:(36) Where,
- Corresponding Input
(1) (2) (3) (4) (5) (6) (7) (8) (9) (10) /MAT/LAW6/mat_ID/unit_ID or /MAT/HYDRO/mat_ID/unit_ID AbsolutePRESSURE_AbsoluteENERGY 0 0 0 0 0 0 C4 = 1 C5 = - Output Results
Table 3. Time History Measure Initial Value Unit /TH/BRICK (P) P P0 Pressure /TH (IE) Energy /TH/BRICK (IE) Pressure - Comparison with Theoretical ResultNumerical result for perfect gas pressure is given by time history. Element time history (/TH/BRICK) allows displaying it. This result is compared to a theoretical one. Curves are superimposed.
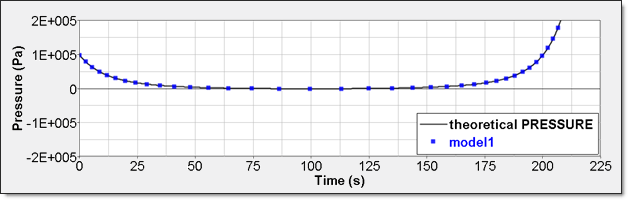
Figure 7. Numerical Pressure, Model 1:Internal energy can be obtained through two different ways. The first one is internal energy density () recorded by element time history (/TH/BRICK). The second one is the internal energy from the global time history because the model is composed of a single element.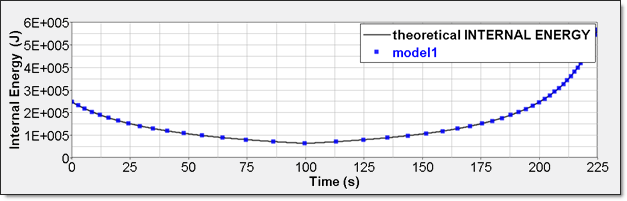
Figure 8. Numerical Internal Energy, Model 1:
Case 2: Pressure is Relative and Energy is Absolute
- Equation of StateEquation of state for a perfect gas is:
(37) Calculating Pressure from a reference one provides relative pressure:(38) Expanding this expression and identifying with polynomial coefficients leads to:(39) Where,
- Minimum Pressure
(40) Then, the minimum pressure must be set to a non-zero value .
- Corresponding Input
(1) (2) (3) (4) (5) (6) (7) (8) (9) (10) /MAT/LAW6/mat_ID/unit_ID or /MAT/HYDRO/mat_ID/unit_ID RelativePRESSURE_AbsoluteENERGY 0 0 0 0 -P0 P0 C4 = C5 = - Output Result
Time History Measure Initial Value Unit /TH/BRICK (P) 0 Pressure /TH (IE) Energy /TH/BRICK (IE) Pressure - Comparison with Theoretical ResultElement time history (/TH/BRICK) is the pressure relative to Psh. The resulting curve is then shifted with Psh value and starts from 0.
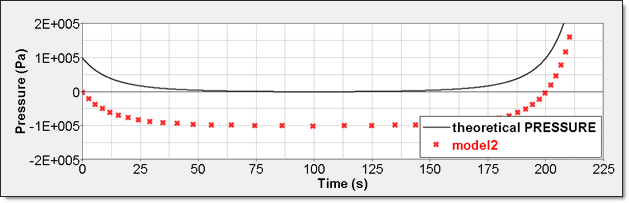
Figure 9. Numerical Pressure, Model 2:Internal energy can be obtained through two different ways. The first one is internal energy density () recorded by element time history (/TH/BRICK). The second one is the internal energy from the global time history because the model is composed of a single element.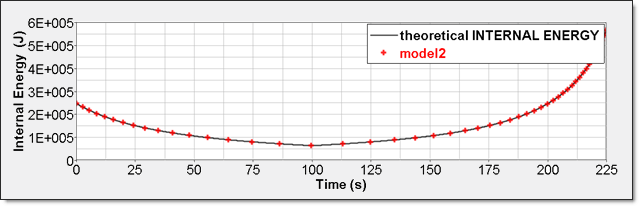
Figure 10. Numerical Internal Energy, Model 2:
Case 3: Both Pressure and Energy are Relative
- Equation of StateEquation of state for a perfect gas is:
(41) Initial internal energy can be introduced:(42) Calculating pressure from a reference one provides:(43) Where,(44) Expanding this expression and identifying with polynomial coefficients leads to:(45) Where, - Minimum Pressure
(46) The minimum pressure must be set to a non-zero value .
- Corresponding Input
(1) (2) (3) (4) (5) (6) (7) (8) (9) (10) /MAT/LAW6/mat_ID/unit_ID or /MAT/HYDRO/mat_ID/unit_ID RelativePRESSURE_RelativeENERGY 0 0 -P0 P0 C4 = C5 = 0 - Output Results
Time History Measure Initial Value Unit /TH/BRICK (P) 0 Pressure /TH (IE) 0 Energy /TH/BRICK (IE) 0 Pressure - Comparison with Theoretical ResultElement time history (/TH/BRICK) is the pressure relative to Psh. The resulting curve is then shifted with Psh value and starts also from 0.
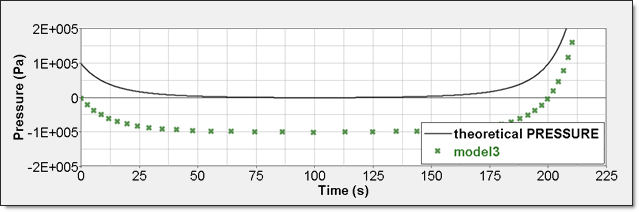
Figure 11. Numerical Pressure, Model 3:Internal energy can be obtained through two different ways. The first one is internal energy density () recorded by element time history (/TH/BRICK). The second one is the internal energy from the global time history because the model is composed of a single element. This numerical internal energy is relative to its initial value; it is shifted with the value from the absolute theoretical one and also starts from 0.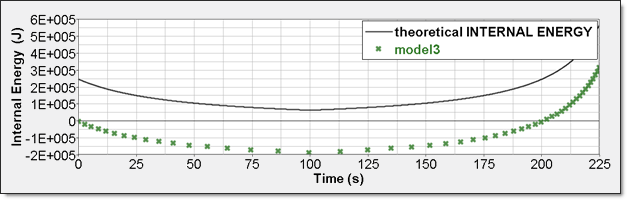
Figure 12. Numerical Internal Energy, Model 3:
Case 4: Pressure is Absolute and Energy is Relative
- Equation of StateEquation of state for a perfect gas is:
(47) Initial internal energy can be introduced:(48) Which leads to:(49) Expanding this expression and identifying with polynomial coefficients leads to:(50) Where, - Corresponding Input
(1) (2) (3) (4) (5) (6) (7) (8) (9) (10) /MAT/LAW6/mat_ID/unit_ID or /MAT/HYDRO/mat_ID/unit_ID AbsolutePRESSURE_RelativeENERGY 0 0 0 0 C4 = C5 = 0 - Output Results
Time History Measure Initial Value Unit /TH/BRICK (P) P P0 Pressure /TH (IE) 0 Energy /TH/BRICK (IE) 0 Pressure - Comparison with Theoretical ResultElement time history (/TH/BRICK) gives absolute pressure. This result is compared to a theoretical one. Curves are superimposed.
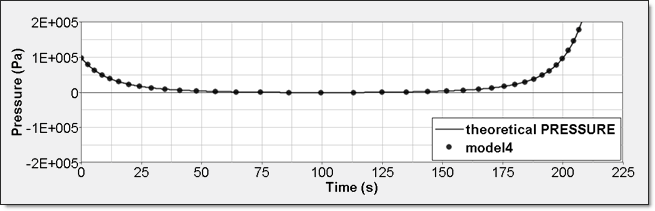
Figure 13. Numerical Pressure, Model 4:Internal energy can be obtained through two different ways. The first one is internal energy density () recorded by element time history (/TH/BRICK). The second one is the internal energy from the global time history because the model is composed of a single element. This numerical internal energy is relative to its initial value; it is shifted with the value from the absolute theoretical one and also starts from 0.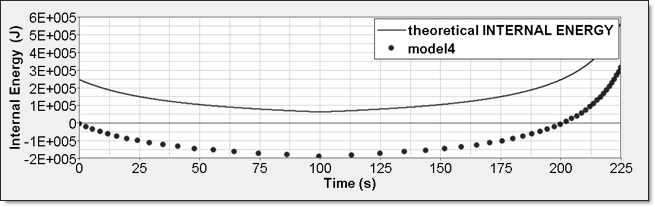
Figure 14. Numerical Internal Energy, Model 4: